Introduction
When sildenafil citrate first entered clinical practice in 1998, it revolutionized the management of erectile dysfunction. The story of a failed antihypertensive drug becoming the world’s most famous “blue pill” is often retold as a triumph of serendipity. Yet, sildenafil’s pharmacology extends far beyond penile hemodynamics. Increasingly, evidence suggests that chronic phosphodiesterase type 5 (PDE5) inhibition has systemic effects, some of which touch the very heart of male reproductive endocrinology.
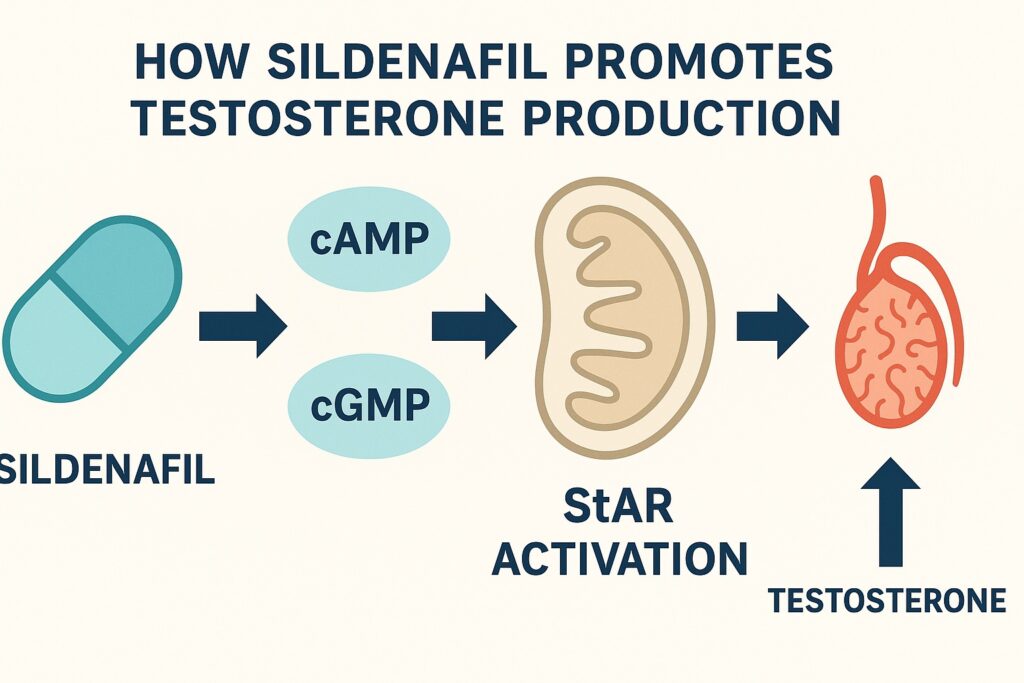
One of the most intriguing discoveries of the past decade is sildenafil’s ability to stimulate testosterone production by Leydig cells, the testicular factories of androgens. This effect is mediated not only by the classic cyclic GMP (cGMP) signaling but also by a synergistic interplay with cyclic AMP (cAMP), the canonical messenger of luteinizing hormone (LH). The result is a fine orchestration that enhances steroidogenesis through modulation of steroidogenic acute regulatory (StAR) protein and related enzymes.
This article unpacks the mechanisms, experimental evidence, and clinical implications of sildenafil’s impact on Leydig cell steroidogenesis, based on detailed in vivo and ex vivo studies in rodent models, and contextualizes the findings within the broader framework of male reproductive medicine.
Leydig Cells and the Physiology of Testosterone Production
Leydig cells, nestled in the interstitial spaces of the testes, are the principal source of testosterone in males. Their function is governed primarily by LH signaling. When LH binds its receptor (LHR), intracellular cAMP levels rise, activating protein kinase A (PKA). PKA phosphorylates proteins that facilitate cholesterol transport into mitochondria — a process mediated by the translocator protein (TSPO) and, crucially, the StAR protein.
Once inside mitochondria, cholesterol undergoes enzymatic transformations through cytochrome P450scc (CYP11A1), hydroxysteroid dehydrogenases, and CYP17A1, ultimately yielding testosterone. This tightly regulated pathway ensures adequate androgen supply for puberty, fertility, libido, and maintenance of secondary sexual characteristics.
While cAMP is the dominant regulator, other signaling cascades intersect. Among them, the nitric oxide (NO)–cGMP–protein kinase G (PKG) axis plays a modulatory role. NO, at low concentrations, stimulates cGMP production, which in turn phosphorylates StAR protein, enhancing steroidogenesis. At high concentrations, paradoxically, NO can inhibit steroidogenic enzymes. Thus, Leydig cell function emerges from a delicate balance of cAMP-driven and cGMP-modulated processes.
Why Sildenafil Matters in the Testis
Sildenafil was designed as a selective PDE5 inhibitor, prolonging cGMP activity by blocking its degradation. While PDE5 is most studied in penile smooth muscle, it is also expressed in testicular tissue, including Leydig cells. By inhibiting PDE5, sildenafil may elevate cGMP levels locally, potentially influencing testosterone biosynthesis.
The study by Andric and colleagues tested this hypothesis through chronic sildenafil administration to adult male rats. Over 30 days, sildenafil-treated animals exhibited significantly higher serum testosterone levels and enhanced ex vivo Leydig cell steroidogenesis. Importantly, these effects persisted even when cells were stimulated with human chorionic gonadotropin (hCG), suggesting that PDE5 inhibition augments the physiological LH–cAMP pathway rather than bypassing it.
This finding carries profound implications: PDE5 inhibitors, while prescribed for erectile dysfunction or pulmonary hypertension, may also reshape endocrine function, possibly benefiting hypogonadal men.
Experimental Evidence: What the Data Show
The key findings from prolonged sildenafil treatment in vivo can be summarized as follows:
- Serum Testosterone: Rats receiving sildenafil (1.25 mg/kg daily) for 30 days demonstrated a moderate but statistically significant increase in serum testosterone compared to controls.
- Leydig Cell Steroidogenesis: Ex vivo cultures of Leydig cells from sildenafil-treated rats produced more testosterone under basal conditions and showed heightened responsiveness to hCG stimulation.
- cAMP and cGMP Dynamics:
- Basal cAMP levels in Leydig cells increased after sildenafil treatment.
- cGMP production was enhanced in response to NO donors, especially when combined with hCG stimulation.
- Expression of cAMP-degrading PDE4A was reduced, explaining the elevated cAMP accumulation.
- Gene Expression: While most steroidogenic enzyme transcripts (e.g., CYP11A1, HSD3B) remained unchanged, CYP17A1 expression increased selectively.
- NO–cGMP Signaling Modulation: Sildenafil downregulated inducible nitric oxide synthase (NOS2) while upregulating guanylyl cyclase subunits (GUCY1A1, GUCY1B1). This shift made Leydig cells more sensitive to physiological NO, enhancing cGMP production without excessive inhibitory effects of high NO.
- StAR Protein Activation: The mature form of StAR protein, essential for cholesterol transport into mitochondria, was more abundant in sildenafil-treated Leydig cells. This correlates directly with enhanced steroidogenic capacity.
Taken together, these findings reveal that sildenafil amplifies testosterone production by orchestrating both cAMP and cGMP signaling, converging on StAR as the pivotal mediator.
Mechanistic Insights: Dual Pathways, One Goal
The elegance of sildenafil’s effect lies in its dual modulation:
- On the cAMP side, sildenafil suppresses PDE4A expression, allowing greater accumulation of cAMP in response to LH/hCG. This potentiates PKA activation and upregulation of steroidogenic genes.
- On the cGMP side, PDE5 inhibition increases Leydig cell sensitivity to NO by upregulating guanylyl cyclase and decreasing cGMP-specific PDE6C. This favors cGMP accumulation, activation of PKG, and phosphorylation of StAR.
The convergence of these two pathways at the level of StAR ensures enhanced cholesterol transport and steroid hormone biosynthesis. This mechanism illustrates a remarkable synergy between canonical and auxiliary second messenger systems.
It is worth noting that while StAR mRNA levels did not significantly change, the protein abundance increased. This discrepancy highlights the importance of post-transcriptional regulation — a reminder that biology rarely behaves as neatly as textbooks suggest.
Potential Clinical Implications
If findings in rats translate to humans, sildenafil could have broader utility than previously imagined. Increased testosterone production might offer adjunctive benefits in conditions such as late-onset hypogonadism, metabolic syndrome, or type 2 diabetes, where low testosterone frequently coexists with erectile dysfunction.
Indeed, small clinical studies have already reported modest increases in serum testosterone in men treated chronically with PDE5 inhibitors. The combined use of testosterone replacement therapy (TRT) with PDE5 inhibitors has also been shown to synergistically improve erectile function in hypogonadal men.
However, enthusiasm should be balanced with caution. Elevated testosterone is not universally beneficial: prostate health, cardiovascular safety, and metabolic effects must all be carefully monitored. Moreover, the rat model employed relatively low sildenafil doses compared with human therapeutic ranges; extrapolation requires rigorous clinical validation.
The Broader Biological Significance
Beyond its clinical potential, this line of research underscores an important principle in pharmacology: drugs are rarely as selective as we imagine. By targeting PDE5, sildenafil not only sustains penile erection but also rewires endocrine signaling at the testicular level.
Such pleiotropic effects invite further exploration. Could chronic PDE5 inhibition improve spermatogenesis by optimizing androgenic support within the seminiferous tubules? Might it protect Leydig cells from apoptosis, as observed in diabetic models? Could testicular blood flow be another mechanism, as preliminary data suggest?
Each of these questions extends the relevance of sildenafil from the urologist’s office to the broader realm of reproductive endocrinology and even men’s general health.
Limitations and Open Questions
Like all research, the findings must be interpreted within context. Rat Leydig cells, though similar, are not identical to human ones. The dose and duration of sildenafil treatment, the absence of comorbid conditions, and the focus on a relatively narrow set of molecular markers all limit generalizability.
Moreover, the interplay between NO, cGMP, and steroidogenesis is notoriously complex. High NO concentrations inhibit steroidogenic enzymes, raising the possibility that under certain conditions, sildenafil could paradoxically suppress androgen production.
Finally, long-term endocrine consequences — both beneficial and adverse — remain underexplored in clinical populations. Does chronic PDE5 inhibition alter feedback loops within the hypothalamic-pituitary-gonadal axis? Could testosterone increases contribute to cardiovascular risks in predisposed individuals? These questions require careful, longitudinal investigation.
Conclusion
Sildenafil’s story is far from complete. What began as a vascular drug repurposed for erections has now expanded into the realm of reproductive endocrinology. By enhancing cAMP and cGMP signaling in Leydig cells, sildenafil stimulates testosterone biosynthesis through activation of the StAR protein and related machinery.
This discovery not only enriches our understanding of testicular physiology but also opens new horizons for therapeutic exploration. For men with both erectile dysfunction and borderline hypogonadism, PDE5 inhibitors might offer a two-for-one solution. For clinicians, the message is clear: the pharmacological reach of sildenafil extends beyond the penis — right into the testis.
FAQ
1. Does sildenafil increase testosterone in men as well as rats?
Some clinical studies suggest modest increases in serum testosterone during chronic PDE5 inhibitor use. However, evidence is limited and variable, so more large-scale trials are needed.
2. Could sildenafil replace testosterone therapy in hypogonadal men?
Not entirely. While sildenafil may boost endogenous testosterone, men with true primary hypogonadism (testicular failure) generally require direct testosterone replacement. Sildenafil may serve as an adjunct rather than a substitute.
3. Are there risks to higher testosterone from chronic sildenafil use?
Potentially. While higher testosterone can improve libido, energy, and sexual function, it may exacerbate prostate growth or cardiovascular risks in susceptible men. Clinical monitoring is essential.