Introduction
Few drugs in modern medicine have become as iconic as sildenafil citrate. Popularly known as Viagra, this phosphodiesterase type 5 (PDE5) inhibitor has redefined the treatment of erectile dysfunction (ED) and has extended its therapeutic reach into pulmonary arterial hypertension (PAH). Its reputation as a pharmacological game-changer is well deserved. Yet, as with all powerful agents, there is more to sildenafil than meets the eye.
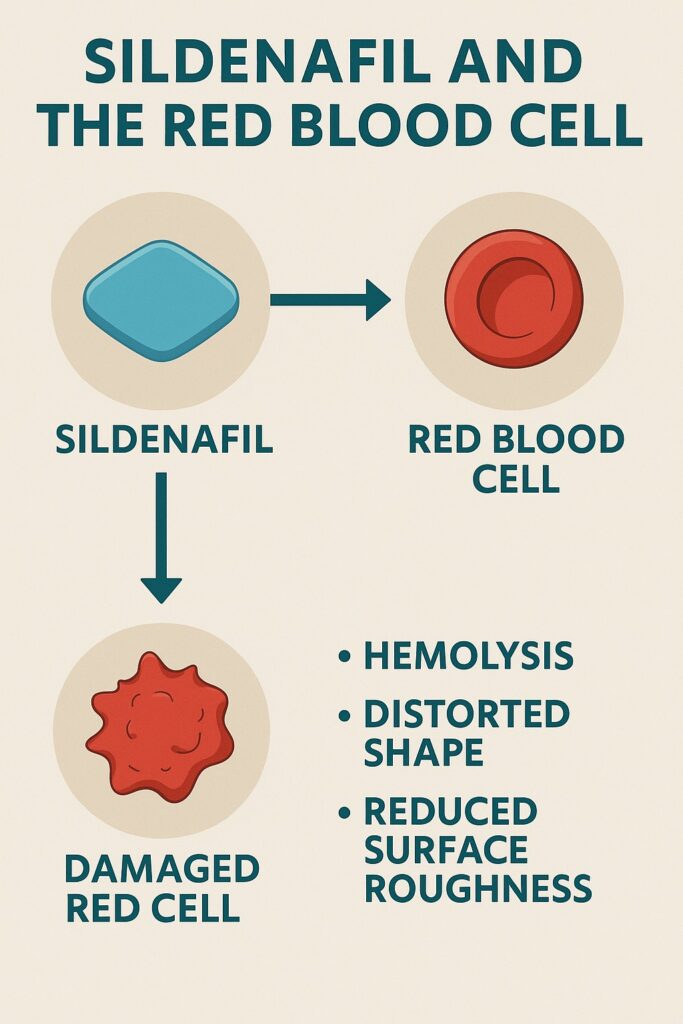
A recent exploration into the drug’s effects on human erythrocytes — the humble red blood cells (RBCs) that faithfully carry oxygen across our tissues — has uncovered unexpected findings. Using atomic force microscopy (AFM), investigators revealed that sildenafil, at supratherapeutic concentrations, can disrupt the ultrastructure of erythrocyte membranes, reduce their surface roughness, and even induce hemolysis.
This article examines the significance of these findings. We will navigate through the methods, results, and interpretations of AFM-based research on sildenafil, and then ask what such cellular-level observations mean for patients, clinicians, and scientists alike. While erythrocytes are not the intended pharmacological target of sildenafil, their vulnerability offers a window into both the elegance and risks of drug-cell interactions.
Sildenafil: Beyond the Penis and Pulmonary Arteries
Sildenafil’s success lies in its elegant hijacking of the nitric oxide (NO)–cyclic guanosine monophosphate (cGMP) signaling cascade. By competitively inhibiting PDE5, sildenafil allows cGMP to accumulate, promoting smooth muscle relaxation and vasodilation. In the penis, this translates into erection; in the pulmonary vasculature, into relief of pressure and strain.
But drugs rarely remain confined to their chosen tissues. After intestinal absorption, sildenafil enters systemic circulation, where it encounters trillions of erythrocytes. These cells, though lacking nuclei, are hardly inert. Their biconcave membranes, decorated with proteins and lipids, provide potential docking sites for xenobiotics. A drug that binds, embeds, or perturbs this delicate membrane could alter cellular stability.
Why does this matter? Red blood cells are not only oxygen couriers but also critical players in microcirculatory flow. Their deformability, surface charge, and membrane roughness determine how well they squeeze through capillaries, exchange gases, and resist shear stress. Disturb these properties, and systemic consequences may follow.
The Power of Atomic Force Microscopy
Traditional microscopy has taught us much about cells, but AFM pushes the frontier further. Invented in the 1980s, AFM allows researchers to scan surfaces at nanometer resolution, detecting the minute undulations of biological membranes. Unlike electron microscopy, AFM operates in near-physiological conditions, providing real-time, three-dimensional surface maps.
Applied to erythrocytes, AFM uncovers their signature membrane patterns: holes, blebs, and corrugations that reflect underlying cytoskeletal architecture. Quantitative parameters such as root-mean-square (RMS) surface roughness can be extracted, offering a measure of how “textured” or “smooth” the cell surface appears. Changes in roughness often indicate alterations in protein organization, lipid fluidity, or cytoskeletal integrity.
Thus, when AFM shows that sildenafil reduces RBC membrane roughness, this is not merely an aesthetic observation. It implies disruption of the cell’s cytoarchitecture, with potential consequences for mechanical resilience.
Study Overview: Sildenafil Meets the Red Cell
The AFM study in question incubated normal human erythrocytes with sildenafil citrate at a concentration of 2.5 mg/mL — significantly higher than clinical dosing, but chosen deliberately to exaggerate and detect membrane effects. The cells were examined after up to 90 minutes of exposure.
Several striking findings emerged:
- Hemolysis at high concentration: After incubation, red-colored supernatants signaled the rupture of RBCs and release of hemoglobin.
- Distorted morphology: Normally biconcave cells shrank and lost their disk-like elegance, leaving behind smaller, irregular “ghosts.”
- Loss of membrane pattern: The characteristic holes and blebs disappeared, replaced by granular structures suggestive of protein denaturation.
- Reduced roughness: RMS surface roughness dropped significantly, from ~23 nm in controls to ~15 nm after sildenafil exposure.
In short, sildenafil transformed robust erythrocytes into fragile remnants, stripped of their ultrastructural identity.
What Does Reduced Surface Roughness Mean?
At first glance, a smoother cell membrane may sound beneficial — after all, smooth is often equated with streamlined. Yet, in biology, roughness is a sign of vitality. The undulating architecture of an RBC membrane reflects the anchoring of spectrin-actin cytoskeletal networks to integral proteins. These protrusions and depressions are not random noise; they are the signatures of resilience.
When roughness diminishes, it often signals that anchoring proteins are disengaged, the cytoskeleton is compromised, and the membrane has lost its mechanical tension. The result is a cell that is less deformable, more fragile, and more prone to rupture under stress.
Therefore, sildenafil’s reduction of RBC roughness hints at its interference with membrane proteins or lipids. Whether this interaction occurs via direct binding, lipid bilayer integration, or disruption of protein complexes remains unclear. But the outcome — hemolysis — speaks volumes.
Hemolysis in Context: Laboratory vs. Life
Should patients worry that their Viagra tablet is silently shredding their erythrocytes? Not necessarily. The AFM study used concentrations several-fold higher than therapeutic plasma levels. In clinical practice, sildenafil is generally well tolerated, with hematologic side effects rarely reported.
However, the findings are not trivial. They highlight the principle that drugs can exert off-target, membrane-level actions independent of their canonical receptors. For most healthy individuals, these effects may be negligible. But in certain contexts — chronic overdose, recreational abuse, or preexisting fragility of erythrocytes (as in hemoglobinopathies or anemia) — the membrane-destabilizing potential of sildenafil may contribute to clinical problems.
In other words, while the average patient is safe, the research urges caution in those at the margins.
Comparisons With Other Drugs and Agents
The AFM observations of sildenafil join a growing literature on drug-induced erythrocyte alterations. Local anesthetics like procaine, asthma medications, and even heavy metals like aluminum and zinc have been shown to alter RBC morphology and roughness. Some cause echinocyte formation, others spherocytosis, and many reduce membrane resilience.
This body of evidence underscores the vulnerability of erythrocytes to chemical insults. They may lack nuclei, but their membranes are chemically dynamic, responsive, and fragile. As such, erythrocytes often serve as “canaries in the coal mine,” signaling toxic interactions long before organ-level dysfunction is evident.
Clinical Implications: Should We Care?
The immediate takeaway is not to alarm patients but to inform clinicians. Sildenafil remains safe within prescribed doses. But three key implications emerge:
- Caution in vulnerable populations: Patients with anemia, sickle cell disease, or thalassemia may be more susceptible to membrane disruption.
- Abuse risk: Recreational users who consume large, repeated doses may unknowingly risk erythrocyte fragility.
- Drug development insight: AFM-based erythrocyte testing could serve as an early screen for membrane toxicity of new drugs, complementing standard pharmacology assays.
These implications highlight the dual role of such research: it refines clinical vigilance while advancing methodological innovation.
A Window Into the Future: AFM as a Diagnostic Tool
Beyond its role in research, AFM holds potential as a diagnostic adjunct. Subtle alterations in RBC membrane roughness could serve as biomarkers for disease or drug exposure. Smokers, for example, exhibit reduced RBC roughness compared with nonsmokers. Heavy metal exposure and systemic intoxication leave their signatures on cell membranes.
Thus, AFM may become more than a research toy. In the future, clinicians could use nanoscale scanning to assess the hidden impact of medications, toxins, or diseases on the blood. Sildenafil’s story, then, is not merely about one drug but about a paradigm shift in how we visualize pharmacology at the cellular level.
Conclusion
Sildenafil citrate has earned its fame as a PDE5 inhibitor that restores function and relieves suffering. Yet, as the atomic force microscope reveals, its influence extends to unsuspecting targets — red blood cells. At high concentrations, sildenafil smooths their membranes, distorts their shapes, and induces hemolysis.
For the average patient, these effects remain theoretical curiosities, buffered by safe dosing regimens. But for scientists, they offer a cautionary tale and an opportunity. They remind us that every drug, however selective, is ultimately promiscuous at the cellular level. And they invite us to peer deeper — not only into the bloodstream but into the very textures of life itself.
FAQ
1. Does Viagra damage red blood cells in real-life patients?
No convincing evidence suggests clinically relevant erythrocyte damage at therapeutic doses. The AFM findings occurred at much higher concentrations than what is achieved during standard treatment.
2. Why study red blood cells if they are not sildenafil’s main target?
Because erythrocytes are abundant, accessible, and sensitive indicators of membrane toxicity. Observing how a drug alters their ultrastructure can reveal off-target effects and broaden understanding of drug safety.
3. Should patients with anemia or blood disorders avoid sildenafil?
Not categorically. However, caution and medical supervision are advisable in such patients, especially if other risk factors are present. Individualized risk assessment remains the best approach.


